DOJ Moves FDA-Approved and State-Licensed Medical Marijuana to Schedule III, Sets June 29 Hearing on Broader Reclassification
Acting Attorney General Todd Blanche signed an order on April 23 placing FDA-approved cannabinoid drugs and state-licensed medical marijuana in Schedule III — the same tier as ketamine and anabolic steroids — while leaving recreational marijuana in Schedule I pending a June 29 DEA hearing on broader rescheduling.
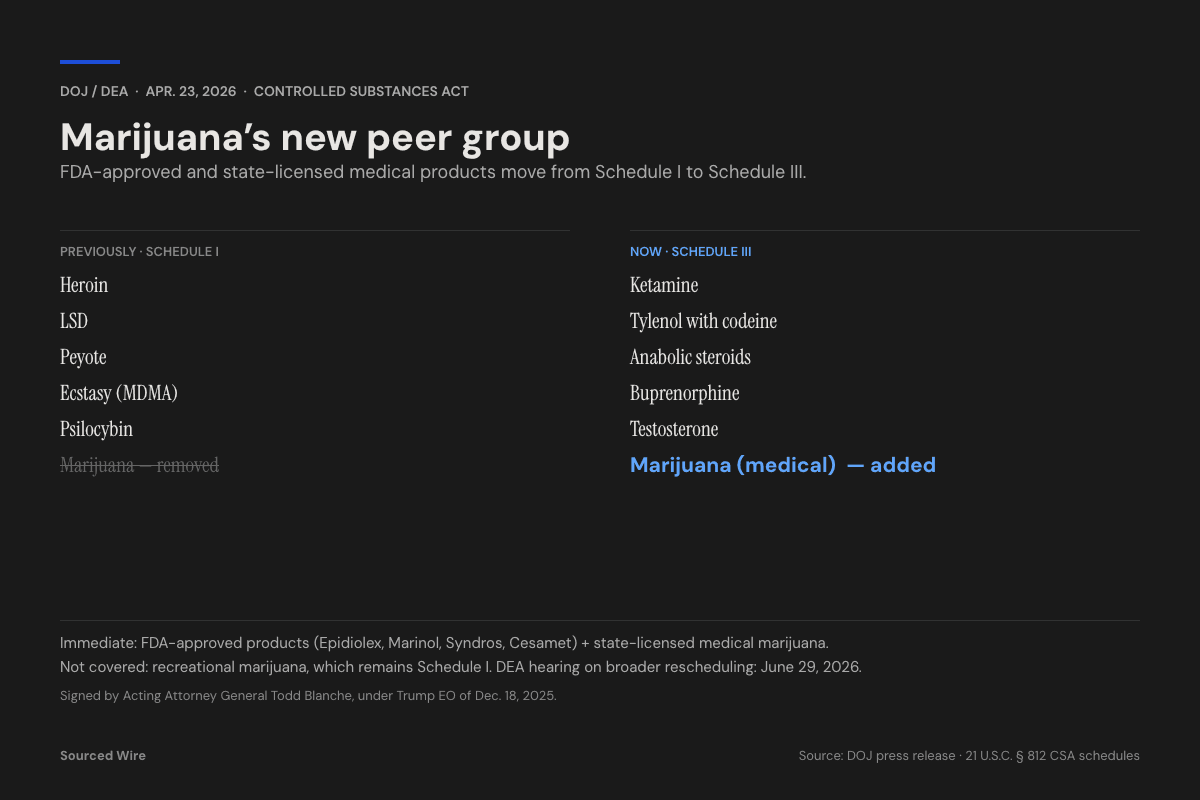
The US Department of Justice and the Drug Enforcement Administration announced on April 23, 2026 that two categories of marijuana products have been immediately rescheduled from Schedule I to Schedule III of the Controlled Substances Act. The order was signed by Acting Attorney General Todd Blanche under a December 18, 2025 executive order from President Trump titled Increasing Medical Marijuana and Cannabidiol Research.
What was rescheduled
The April 23 order applies to exactly two categories of product:
- FDA-approved marijuana products. This covers the four cannabinoid drugs currently on the US market: Epidiolex (cannabidiol, for seizure disorders); Marinol and Syndros (dronabinol, a synthetic THC for chemotherapy-associated nausea and AIDS-related anorexia); and Cesamet (nabilone, also a synthetic cannabinoid).
- State-licensed medical marijuana products. The order establishes an expedited federal registration pathway under which state medical-marijuana licensees may submit their existing state credentials as conclusive evidence of state-law authorization when applying for DEA registration as manufacturers, distributors, or dispensers.
What was not rescheduled
Recreational marijuana remains Schedule I, alongside heroin and LSD, pending the outcome of a new administrative hearing the DEA has scheduled to begin June 29, 2026. That hearing will consider the broader rescheduling of all marijuana to Schedule III — the action Trump's December executive order directed the Attorney General to complete "in the most expeditious manner in accordance with Federal law."
The peer group the drug just joined
Schedule III is defined under 21 U.S.C. § 812 as containing substances with "a potential for abuse less than the drugs or other substances in Schedules I and II," a "currently accepted medical use in treatment in the United States," and a risk of only "moderate or low physical dependence or high psychological dependence."
Medical marijuana now sits alongside:
- Ketamine (dissociative anesthetic, approved depression treatment)
- Buprenorphine (opioid-use-disorder medication)
- Testosterone and other anabolic steroids
- Tylenol with codeine and other combination codeine products
- Nabilone (Cesamet) — already in Schedule II until today's order
The practical consequences are real but narrow:
- Medical-marijuana researchers no longer need a Schedule I license — historically a heavy regulatory lift — to conduct clinical work.
- Schedule III businesses can deduct ordinary business expenses for federal tax purposes, reversing the effect of Internal Revenue Code § 280E, which has barred Schedule I and II marijuana operators from taking standard deductions.
- State-licensed dispensaries still face a dense federal–state regulatory interface, and 280E still applies to recreational sales, which remain Schedule I.
What the June 29 hearing will decide
The broader rescheduling rulemaking — initiated under the Biden administration in 2024 and stalled in administrative-law-judge proceedings through most of 2025 — has now been restarted on an expedited track. The June 29 hearing will build the administrative record for a final rule that would move recreational marijuana and all other marijuana preparations to Schedule III. A Schedule III reclassification of the entire plant would, in effect, collapse much of the distance between federal drug law and the medical- and recreational-use regimes already in place in most US states.
The US cannabis industry generated roughly $47 billion in legal sales in 2025, according to industry tracker BDSA.