Trump EO Orders FDA-DEA Pathway for Ibogaine Access, $50M for State Psychedelic Research
President Trump signed an executive order directing the FDA and DEA to create a Right to Try pathway for psychedelic drugs — explicitly naming ibogaine — and allocating $50 million in HHS funds to match state psychedelic research programs.
President Trump signed an executive order Saturday directing the FDA and DEA to create a Right to Try pathway for psychedelic drugs — naming ibogaine specifically — and allocating $50 million in HHS funding to match state governments investing in psychedelic research programs.
The order, titled "Accelerating Medical Treatments for Serious Mental Illness," is the first federal action to explicitly name ibogaine as a target for expanded patient access. Ibogaine, derived from the West African iboga shrub, is a Schedule I controlled substance in the United States but has been used in unregulated clinics in Mexico to treat opioid dependence. It has also been linked to cardiac deaths.
What the order directs
- FDA Commissioner to issue "Commissioner's National Priority Vouchers" for psychedelic drugs that have received Breakthrough Therapy designation for treating serious mental illness
- FDA and DEA to "establish a pathway for eligible patients to access psychedelic drugs, including ibogaine compounds, under the Right to Try Act," including any Schedule I handling authorizations needed for treating physicians and researchers
- HHS to allocate "at least $50 million from existing funds" through the Advanced Research Projects Agency for Health (ARPA-H) to match state-level psychedelic research programs
- HHS, FDA, and VA to coordinate on clinical trial participation, data sharing, and real-world evidence generation
- Attorney General to "initiate and complete review" of Schedule I substances that complete Phase 3 trials, enabling rescheduling "as quickly as practicable"
The ibogaine detail
The explicit mention of ibogaine compounds in the Right to Try directive is the order's most novel provision. The 2018 Right to Try Act allows terminally ill patients to access drugs that have passed FDA Phase 1 trials but have not yet received full approval. It has rarely been used for psychedelics because most compounds remain on Schedule I, which creates separate DEA handling barriers on top of FDA clearance. The order instructs both agencies to create a unified pathway, including authorizations for physicians and researchers to handle the substance.
No ibogaine product currently holds a Breakthrough Therapy designation. Atai Life Sciences is running a Phase 2 trial of an ibogaine analogue for opioid use disorder. Compass Pathways is in Phase 3 with psilocybin for treatment-resistant depression, which does hold Breakthrough status.
The funding mechanism
The $50 million ARPA-H allocation is a matching-funds structure. States that commit their own money to psychedelic research programs will have those dollars matched from the existing ARPA-H budget — no new appropriation is required. ARPA-H was established in 2022 as a DARPA-style research agency within HHS.
The order frames the funding push as a response to the veteran suicide rate, which it puts at "more than twice" the non-veteran adult rate, with over 6,000 veteran suicides annually for the past 20 years. Research at Stanford, Harvard, and Johns Hopkins is cited as the basis for the policy.
Market reaction preceded the signing
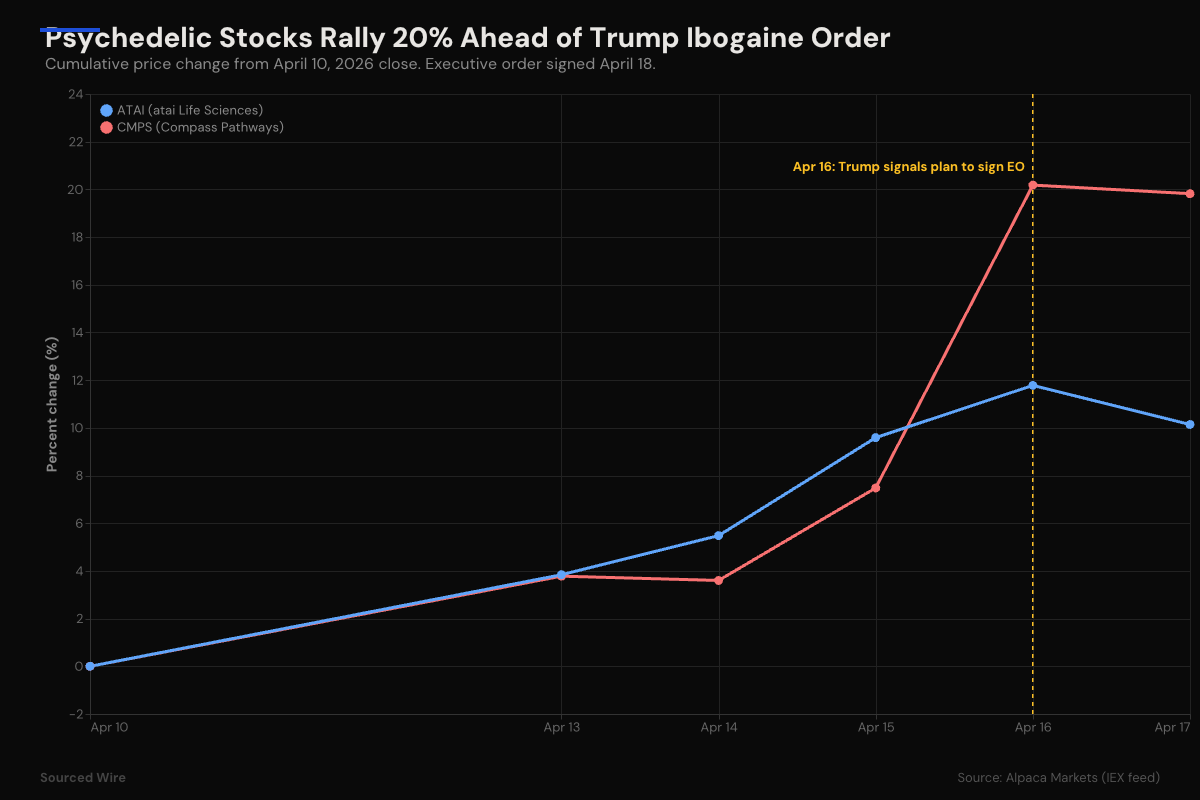
Publicly traded psychedelic companies rallied sharply before the signing. Compass Pathways (CMPS) closed up 11.8% on April 16 after Trump signaled he would sign the order, and ended the week up 19.8% from the April 10 close. atai Life Sciences (ATAI) closed up 2% on April 16 and up 10.1% on the week.
Both stocks sold off slightly on April 17 as traders took profits. Markets were closed April 18, the day of the actual signing.
What happens next
The order sets no specific deadline for the FDA-DEA access pathway to be operational. The Attorney General's rescheduling review is triggered by Phase 3 completion of any Schedule I psychedelic — meaning psilocybin could be the first candidate if Compass Pathways' ongoing Phase 3 trials succeed. MDMA-assisted therapy, which received a Complete Response Letter from the FDA in August 2024, would need to resubmit its application to benefit from the voucher provision.
The FDA's Commissioner's National Priority Voucher program, launched in 2025, accelerates review of drug applications deemed to serve national priorities. It is a separate track from the existing Breakthrough Therapy designation, which already applies to some psychedelic compounds.